
Introduction
Ultra-precision machining for bone screws uses tightly controlled CNC-based operations to produce implantable fasteners with micron-level tolerances and biocompatible surface finishes.
For medical device engineers, orthopedic OEMs, and procurement managers, the stakes are direct: a dimensionally out-of-spec bone screw can fail under load, impair osseointegration, or cause implant-to-instrument mismatch during surgery. Small deviations in thread pitch or minor diameter translate immediately into reduced pullout strength.
This guide covers the full machining process step by step — from material selection and thread geometry to the quality systems that separate reliable manufacturers from those treating bone screws like commodity parts. Quality has to be built into the process, not added at final inspection.
TL;DR
- Ultra-precision machining produces bone screws to tolerances as tight as ±0.0001 in with surface finishes as fine as 4 µin Ra
- Material choice (titanium, stainless steel, cobalt-chromium) dictates tooling grades and machining strategies
- Cortical and cancellous screws require distinct machining approaches due to different thread geometries
- Surface finish governs osseointegration, corrosion resistance, and regulatory acceptance
- Quality control depends on in-process gauging and IQ/OQ/PQ validation at every production stage, not only at final inspection
What Is Ultra-Precision Machining for Bone Screws?
Ultra-precision machining for bone screws is the controlled removal of material from implant-grade metal bar stock to produce threaded fasteners with sub-ten-micron dimensional tolerances and clinically acceptable surface integrity. The finished screw must satisfy two performance specs simultaneously: a mechanical spec (thread form, pitch, minor and major diameter, head geometry) and a biological spec (biocompatibility, surface condition for bone adhesion or removal).
That dual requirement sets it apart from standard CNC screw production in several concrete ways:
- Implant-grade material certifications required for every bar stock lot
- Full traceability from raw material through finished part
- Post-machining passivation to meet biocompatibility standards
- Regulatory process validation (FDA 21 CFR Part 820 / ISO 13485) before production release
Commercial CNC work typically holds ±0.001 in tolerances. Bone screws demand ±0.0002 in or tighter on critical features — a fivefold increase in precision that affects tooling selection, cutting parameters, and inspection protocols at every stage.
Why Ultra-Precision Machining Is Required for Bone Screws
Bone screws operate inside the human body under cyclic mechanical loads. Dimensional deviations in thread pitch, minor diameter, or concentricity translate directly into reduced pullout strength, fatigue failure, or mismatch with mating implant components. Research shows that pullout strength is controlled by major diameter, thread engagement length, and thread shape factor, making dimensional precision non-negotiable.
Regulatory requirements (FDA 21 CFR Part 820, ISO 13485) mandate documented process control and traceability for implantable devices. The FDA's Quality Management System Regulation (QMSR), effective February 2, 2026, incorporates ISO 13485:2016 by reference, aligning US requirements with international standards. Machining processes must be validated before production, not inspected after—process qualification is a regulatory requirement, not a best practice.
The materials used (titanium, Ti-6Al-4V ELI, 316LVM stainless steel, cobalt-chromium) are notoriously difficult to machine. They generate heat, resist cutting, and work-harden rapidly. Cobalt-chromium alloys exhibit high strength, toughness, and low thermal conductivity, resulting in rapid tool wear. Ti-6Al-4V ELI machining is dominated by adhesion and notching wear mechanisms, with poor thermal conductivity preventing rapid heat dissipation. Controlling these variables requires purpose-built tooling geometry, optimized cutting parameters, and active coolant management—not just close tolerances in the CAM file.
Mastering these material challenges pays off at scale. The global orthopedic implants market was valued at USD 26.97 billion in 2025 and is projected to reach USD 36.63 billion by 2033, growing at 3.94% CAGR. Suppliers who can hold tight tolerances on difficult materials—consistently, across production volumes—are positioned to capture a meaningful share of that growth.
These four pressures—mechanical performance, regulatory compliance, material difficulty, and market scale—each independently justify precision manufacturing discipline. Together, they make it the baseline for any credible bone screw supplier.
How the Bone Screw Ultra-Precision Machining Process Works
The process converts certified implant-grade bar stock into a finished, inspection-ready bone screw through a sequence of turning, threading, hole-making, finishing, and cleaning operations. Most of these must be completed in as few setups as possible to preserve concentricity and dimensional integrity.
Single-setup or reduced-setup machining — achieved via multi-spindle screw machines or Swiss-type lathes — is preferred because each re-clamping introduces runout error that accumulates across critical features like the cannula bore, thread form, and head recess.
Maintaining coaxial alignment between those features is essential for both mechanical performance and surgical instrumentation compatibility.
Step 1 — Material Selection and Bar Stock Preparation
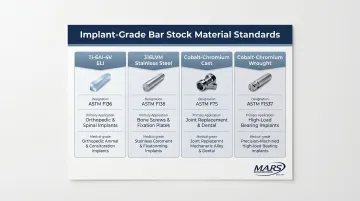
Bone screws begin as certified bar stock (titanium, stainless steel, or cobalt-chromium alloys) that must conform to ASTM or ISO material standards. Common specifications include:
| Alloy Type | Standard | Application |
|---|---|---|
| Ti-6Al-4V ELI | ASTM F136 | Wrought annealed titanium alloy for surgical implants |
| 316LVM Stainless Steel | ASTM F138 | Wrought stainless steel bar and wire for surgical implants |
| Cobalt-Chromium (Cast) | ASTM F75 | Cobalt-chromium-molybdenum casting alloy |
| Cobalt-Chromium (Wrought) | ASTM F1537 | Wrought cobalt-chromium-molybdenum alloys |
Manufacturers must maintain mill certificates and lot traceability from raw material through finished part. Bar stock preparation includes cutting to length, verifying incoming dimensions, and confirming material certification before loading into the screw machine. Shops should specify cold-worked bar stock — most implant alloys cannot be heat treated post-machining to restore strength.
Step 2 — Core Machining (Turning, Threading, and Feature Generation)
The turning stage uses CNC lathes or screw machines to rotate bar stock while cutting tools remove material to establish the screw's outer diameter, head geometry, tip form, and any steps or tapers. For cannulated screws, the bore is drilled and reamed during this stage to maintain concentricity before threading begins.
The threading operation increasingly relies on thread whirling—a single-pass method using a rotating ring of inserts offset from the part centerline. Compared to traditional die-chasing, thread whirling offers several advantages for orthopedic screw production:
- Produces the full thread profile in one pass, eliminating multi-pass chatter
- Minimizes heat generation, protecting tight tolerances on slender screw shafts
- Improves chip evacuation on long bone screw geometries prone to thermal distortion
- Reduces cycle time per part on high-volume medical programs
Shops running multi-spindle CNC screw machines — capable of up to 16 simultaneous operations — can execute turning, drilling, threading, and parting in a single work cycle, cutting cycle time and per-part cost significantly on high-volume medical programs. M.A.R.'s Engineering runs both multi-spindle and single-spindle CNC screw machines alongside in-house Wire EDM, which means custom tooling is produced on-demand rather than waiting weeks for external vendors.
Step 3 — Surface Finishing, Passivation, and Cleaning
Post-machining surface finishing begins with passivation, which uses nitric or citric acid baths per ASTM A967 or ASTM F86 to remove free iron and contaminants from the metal surface. The result is a chromium oxide passive layer that enhances corrosion resistance — mandatory for stainless steel implants and strongly recommended for titanium.
Ultrasonic cleaning and inspection-grade protocols then remove machining fluids, chips, and particulates before final inspection. Residual contamination compromises biocompatibility testing and downstream sterilization — regulatory results need to reflect the implant's actual performance, not surface debris.
Key Factors That Affect Bone Screw Machining Quality
Material Machinability
Titanium alloys generate heat and work-harden at the cutting zone. Stainless steel produces long, stringy chips that risk re-cutting and surface damage. Cobalt-chromium is extremely abrasive on tooling. Each material demands specific cutting speeds, feed rates, coolant strategies, and tool grades to avoid surface defects or dimensional drift.
Thread Geometry and Screw Type
Cortical screws require tight-pitch, shallow threads held to close tolerances across the full thread length. Cancellous screws require deeper, wider-pitch threads that engage soft bone effectively. ISO 5835 specifies that cortical screws (Code HA) have shallow threads, while cancellous screws (Code HB) have deep threads — for example, an HA 4 screw has a pitch of 1.5 mm versus 1.75 mm for an HB 4.
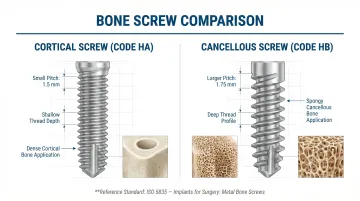
Machining parameters, tooling, and fixturing differ significantly between screw types. Self-tapping tips add further complexity, requiring milling of cutting flutes, thread whirling, and deburring to prevent burr formation in thread roots.
Tooling Condition and Management
Worn inserts don't just reduce part quality — they introduce subsurface stress, microburrs in thread roots, and heat damage that may not appear on inspection but can initiate fatigue cracks in service. Tooling change intervals must be defined and enforced before problems develop, not after.
In-house tooling capability matters here. Wire EDM for custom inserts, for example, cuts replacement tooling lead time from weeks to hours — a meaningful advantage when production schedules can't wait on outside vendors.
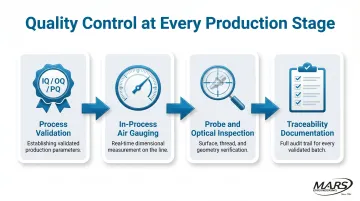
Process Validation and In-Process Measurement
FDA 21 CFR 820.75 requires process validation when results cannot be fully verified by subsequent inspection. IQ/OQ/PQ validation proves that a machining process consistently produces conforming parts across shifts and batches — not just on a good day with a fresh setup.
In-process gauging closes the loop between validation and production. Air gauging, probe-based measurement, and optical inspection detect dimensional drift before it produces a non-conforming lot. Any medical device machining supplier should have documented traceability and ISO-compliant quality systems in place as a minimum operating condition.
Common Issues and Misconceptions in Bone Screw Machining
Misconception: Standard CNC Tolerances Are Sufficient for Bone Screws
Commercial CNC machining typically operates at ±0.005 in to ±0.010 in tolerances, whereas bone screws often require ±0.0002 in or tighter on critical features like the minor diameter and cannula bore. Engineers specifying bone screws without accounting for this gap risk accepting parts that pass visual inspection but fail functional or regulatory requirements.
Misconception: 3D Metal Printing Can Replace Machining for Bone Screws
While additive manufacturing enables surface porosity for osseointegration, it cannot cold-work the material—and most implant alloys derive their strength from cold working during bar stock production. AM devices exhibit anisotropy relative to the build direction, which affects final mechanical properties and dimensional tolerances.
Post-processing heat treatments like Hot Isostatic Pressing (HIP) can reduce residual porosity, but they also reduce the modulus and yield strength of the material. Machining from bar stock remains the baseline process, with additive techniques used selectively for porous coatings or structural geometry not achievable subtractively.
Misconception: Surface Finish Is a Cosmetic Concern
Ra value on a bone screw surface directly affects osseointegration rate, bacterial adhesion risk, and corrosion behavior in vivo. Too smooth a finish reduces bone cell attachment; too rough a finish concentrates stress at surface irregularities. The target finish must be specified by design engineers and verified on every production lot.
Conclusion
Ultra-precision machining of bone screws is a tightly controlled, multi-step manufacturing process where material choice, machining strategy, surface finishing, and quality validation each play a critical role in producing an implant that performs reliably in the body. Every feature—thread form, cannula bore, head recess—must meet both mechanical and biological performance specifications.
Choosing a machining partner who understands the clinical context of bone screw design, and who covers screw forming through finishing and quality documentation under one roof, is as important as the machining process itself.
For medical OEMs evaluating single-source manufacturers, M.A.R.'s Engineering offers ISO-compliant precision screw machining, in-house finishing, and near-zero-lead-time tooling — all in one facility. That means fewer handoffs, faster validation cycles, and a single traceability chain from raw bar stock to finished implant component.
Frequently Asked Questions
How are bone screws made?
Bone screws are machined from certified implant-grade bar stock (titanium, stainless steel, or cobalt-chromium) using CNC turning, thread whirling or single-pass threading, drilling, and surface finishing operations. Parts are then cleaned, passivated, and inspected to tight tolerances before use in surgery.
What screws are used in bone surgery?
The two primary types are cortical screws (for dense bone, with small pitch and shallow threads) and cancellous screws (for spongy bone, with larger pitch and deeper threads). Specialty designs include cannulated, self-tapping, and self-drilling variants, each machined to different specifications.
Why are medical screws so expensive?
Bone screws require implant-grade certified materials, ultra-tight dimensional tolerances, validated manufacturing processes, regulatory documentation, and post-machining finishing, all of which add cost at every stage. Liability exposure drives strict quality control throughout production, and material certifications must be maintained from bar stock through final inspection.
What tolerances are required for bone screw machining?
Critical features such as minor diameter, major diameter, and cannula bore typically require tolerances of ±0.0002 in or tighter, with surface finishes often specified in the range of 8–32 µin Ra depending on the design intent and bone contact interface.
What is thread whirling and why is it preferred for bone screws?
Thread whirling is a single-pass threading process using a rotating ring of inserts that cuts the thread form in one motion with minimal heat, superior chip evacuation, and high accuracy. This makes it the preferred method for long, slender orthopedic screws, where multi-pass threading would introduce chatter or thermal distortion.
What surface finish does a bone screw need to be implant-ready?
The required surface finish depends on the implant's clinical function. Screws intended for osseointegration benefit from a controlled roughness that promotes bone cell adhesion, while all implants must undergo passivation and cleaning to remove contaminants and achieve the corrosion-resistant passive layer required for biocompatibility.


